Nanosonics AuditPro™ uniquely sits with the ultrasound console at point of use, educating the user and enabling consistent incorporation of infection control considerations, as part of every day clinician care.
A point of care solution
Nanosonics AuditPro uniquely sits with the ultrasound console and user at the point of use.
Infection prevention documentation is automatically created every time so your clinicians can focus on patient care.

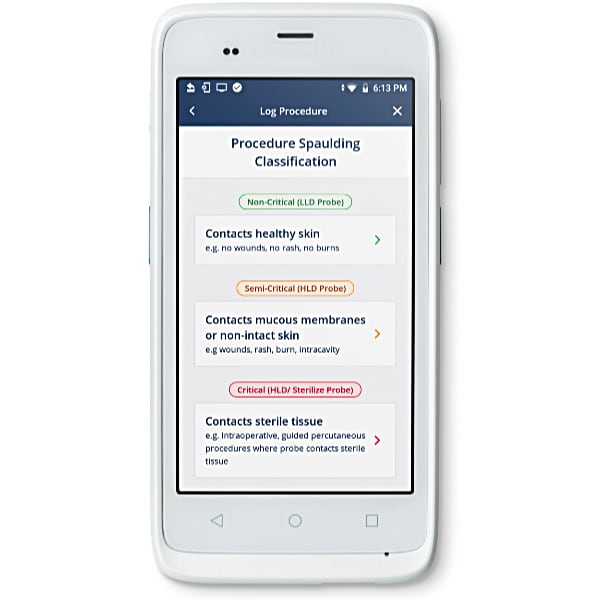
Education through the workflow
Infection prevention education is built in to the Nanosonics AuditPro workflow. Clinical users are guided by the mobile application workflow to qualify every procedure against the Spaulding classification for disinfection requirements.1
Nanosonics AuditPro reduces the burden of ongoing staff retraining, and supports environment with high staff turnover.
Consistent workflow
Every ultrasound user, regardless of experience or department, is guided through the same process to log a procedure to create a digital infection control record to support your standard operating procedure (SOP) every time. This standardization of clinical practice delivers best patient care.

Book your demo of Nanosonics AuditPro
- Spaulding EH (1968). Chemical disinfection of medical and surgical materials. Disinfection, sterilization, and preservation. Lawrence C, Block SS. Philadelphia (PA), Lea & Febiger: 517-531.

